For US Healthcare Professionals
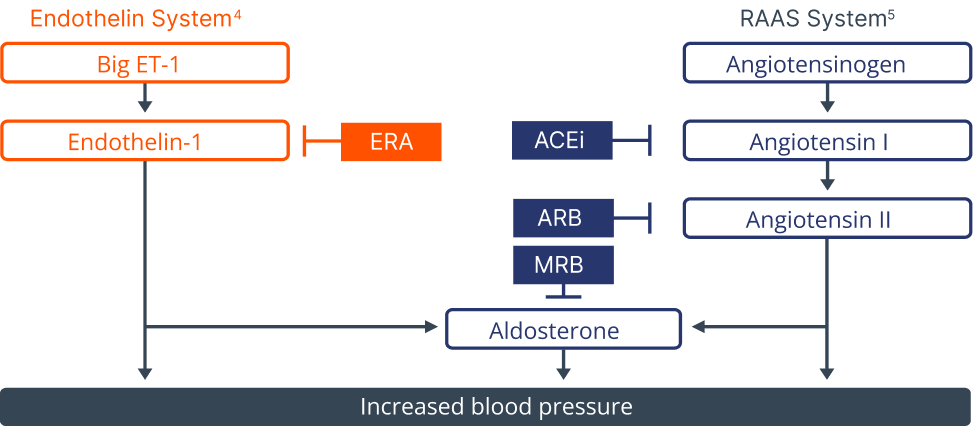
The endothelin system is activated by multiple patient characteristics and comorbiities5

The endothelin system is activated by multiple patient characteristics and comorbidities³
ET-1 acts on smooth muscle cell ETA and ETB receptors to induce contraction, growth, and inflammation1,3,4




BP
158/96 mm Hg
BMI
33.1 kg/m2

Hypertension
CKD Stage 3
Hyperlipidemia

HbA1c: 5.5%
eGFR8: 49 mL/min/1.73 m2
Creatinine: 1.45 mg/dL
Potassium: 5.1 mmol/L

Valsartan 80 mg BID
Amlodipine 10 mg QD
Hydrochlorothiazide 25 mg QD
Atorvastatin 40 mg QD
Dapagliflozin 10 mg QD




BP
154/98 mm Hg
BMI
29.0 kg/m2

Hypertension
Type 2 diabetes mellitus
Hyperlipidemia

HbA1c: 6.8%
eGFR8: 65 mL/min/1.73 m2
Creatinine: 0.9 mg/dL

Valsartan 80 mg BID
Amlodipine 10 mg QD
Hydrochlorothiazide 25 mg QD
Rosuvastatin 20 mg QD
Dapagliflozin 10 mg QD
BID = twice daily; BMI = body mass index; BP = blood pressure; CKD = chronic kidney disease; eGFR = estimated glomerular filtration rate; HbA1c = hemoglobin A1c; QD = once daily.
References: 1. TRYVIO. Prescribing information. Idorsia Pharmaceuticals US Inc; 2025. 2. Schlaich MP, Bellet M, Weber MA, et al. Dual endothelin antagonist aprocitentan for resistant hypertension (PRECISION): a multicentre, blinded, randomised, parallel-group, phase 3 trial. Lancet. 2022;400(10367):1927-1937. Published correction appears in Lancet. 2023;401(10373):268. doi:10.1016/S0140-6736(22)02034-7 3. Davis J, Oparil S. Novel medical treatments for hypertension and related comorbidities. Curr Hypertens Rep. 2018;20(10):90. doi:10.1007/s11906-018-0890-y 4. Dhillon S. Aprocitentan: first approval. Drugs. 2024;84(7):841-847. doi:10.1007/s40265-024-02053-0 5. Heidari Nejad S, Azzam O, Schlaich MP. Dual endothelin antagonism with aprocitentan as a novel therapeutic approach for resistant hypertension. Curr Hypertens Rep. 2023;25(10):343-352. doi:10.1007/s11906-023-01259-z 6. Kostov K. The causal relationship between endothelin-1 and hypertension: focusing on endothelial dysfunction, arterial stiffness, vascular remodeling, and blood pressure regulation. Life (Basel). 2021;11(9):986. doi:10.3390/life11090986 7. Clozel M. Aprocitentan and the endothelin system in resistant hypertension. Can J Physiol Pharmacol. 2022;100(7):573-583. doi:10.1139/cjpp-2022-0010 8. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. Kidney Int Suppl. 2013;3(1):1-150. Accessed July 25, 2025.TRYVIO is contraindicated:
Based on data from animal reproduction studies with endothelin receptor antagonists (ERAs), TRYVIO may cause fetal harm when administered during pregnancy and is contraindicated for use in patients who are pregnant. The available human data for endothelin receptor antagonists do not establish the presence or absence of fetal harm related to the use of TRYVIO. Counsel patients who can become pregnant about the potential risk to a fetus. Obtain a pregnancy test prior to initiation of treatment with TRYVIO. Advise patients who can become pregnant to use effective contraception during treatment, and for one month after the final dose of TRYVIO. When pregnancy is detected, discontinue TRYVIO as soon as possible.
Elevations of aminotransferases and hepatotoxicity are known effects of ERAs, including TRYVIO. Elevations in alanine transaminase (ALT) or aspartate aminotransferase (AST) of greater than 5-fold upper limit of normal (ULN) were observed rarely in patients treated with aprocitentan in the clinical trial, including cases with positive rechallenge. To reduce the risk of potential serious hepatotoxicity, measure serum aminotransferase levels and total bilirubin prior to initiation of treatment and repeat during treatment periodically and as clinically indicated.
Do not initiate TRYVIO in patients with elevated aminotransferases (>3 × ULN) or moderate to severe hepatic impairment. Advise patients with symptoms suggesting hepatotoxicity (nausea, vomiting, right upper quadrant pain, fatigue, anorexia, scleral icterus, jaundice, dark urine, fever, or itching) to immediately stop treatment with TRYVIO and seek medical attention.
If sustained, unexplained, clinically relevant aminotransferase elevations occur, or if elevations are accompanied by an increase in bilirubin >2 × ULN, or if clinical symptoms of hepatotoxicity occur, discontinue TRYVIO.
Fluid retention and peripheral edema are known effects of ERAs, including TRYVIO. Edema/fluid retention was reported in 9% of TRYVIO-treated patients compared with 18% of patients receiving aprocitentan 25 mg (twice the recommended dose) and 2% on placebo in the clinical trial, requiring additional diuretic use in some patients. Older age and chronic kidney disease are risk factors for edema/fluid retention with TRYVIO. TRYVIO has not been studied in patients with heart failure New York Heart Association stage III–IV, unstable cardiac function, or with NTproBNP ≥500 pg/mL. TRYVIO is not recommended in these patients.
Monitor for signs and symptoms of fluid retention, weight gain, and worsening heart failure. If clinically significant fluid retention develops, treat appropriately, and consider discontinuation of TRYVIO.
Decreases in hemoglobin concentration and hematocrit have occurred following administration of other ERAs and were observed in the clinical trial with TRYVIO. Hemoglobin decreases usually presented early, stabilized thereafter, and were reversible after discontinuation. A decrease in hemoglobin of >2 g/dL from baseline was observed in 7% of patients compared to 1% of placebo patients. A decrease to below 10.0 g/dL was observed in 3% of TRYVIO-treated patients compared to 0 patients taking placebo. Initiation of TRYVIO is not recommended in patients with severe anemia. Measure hemoglobin prior to initiation of treatment and periodically during treatment as clinically indicated.
TRYVIO, like other ERAs, may have an adverse effect on spermatogenesis. Counsel men about potential effects on fertility.
The most common adverse reactions (more frequent than placebo and ≥2% in TRYVIO-treated patients) are edema/fluid retention and anemia.
There are no data on the presence of aprocitentan in human milk, the effects on the breastfed infant, or the effect on milk production. Because of the potential for serious adverse reactions in breastfed infants, advise women not to breastfeed during treatment with TRYVIO.
TRYVIO is not recommended in patients with kidney failure (eGFR <15 mL/min) or on dialysis. Patients with renal impairment are at increased risk of edema/fluid retention.
TRYVIO is not recommended in patients with moderate and severe hepatic impairment (Child-Pugh class B and C) because these patients may be at increased risk for poor outcomes from hepatotoxicity.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088. Please see full Prescribing Information including BOXED Warning and Medication Guide.
TRYVIO is an endothelin receptor antagonist indicated for the treatment of hypertension in combination with other antihypertensive drugs, to lower blood pressure in adult patients who are not adequately controlled on other drugs. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions.
TRYVIO is contraindicated:
Based on data from animal reproduction studies with endothelin receptor antagonists (ERAs), TRYVIO may cause fetal harm when administered during pregnancy and is contraindicated for use in patients who are pregnant. The available human data for endothelin receptor antagonists do not establish the presence or absence of fetal harm related to the use of TRYVIO. Counsel patients who can become pregnant about the potential risk to a fetus. Obtain a pregnancy test prior to initiation of treatment with TRYVIO. Advise patients who can become pregnant to use effective contraception during treatment, and for one month after the final dose of TRYVIO. When pregnancy is detected, discontinue TRYVIO as soon as possible.
Elevations of aminotransferases and hepatotoxicity are known effects of ERAs, including TRYVIO. Elevations in alanine transaminase (ALT) or aspartate aminotransferase (AST) of greater than 5-fold upper limit of normal (ULN) were observed rarely in patients treated with aprocitentan in the clinical trial, including cases with positive rechallenge. To reduce the risk of potential serious hepatotoxicity, measure serum aminotransferase levels and total bilirubin prior to initiation of treatment and repeat during treatment periodically and as clinically indicated.
Do not initiate TRYVIO in patients with elevated aminotransferases (>3 × ULN) or moderate to severe hepatic impairment. Advise patients with symptoms suggesting hepatotoxicity (nausea, vomiting, right upper quadrant pain, fatigue, anorexia, scleral icterus, jaundice, dark urine, fever, or itching) to immediately stop treatment with TRYVIO and seek medical attention.
If sustained, unexplained, clinically relevant aminotransferase elevations occur, or if elevations are accompanied by an increase in bilirubin >2 × ULN, or if clinical symptoms of hepatotoxicity occur, discontinue TRYVIO.
Fluid retention and peripheral edema are known effects of ERAs, including TRYVIO. Edema/fluid retention was reported in 9% of TRYVIO-treated patients compared with 18% of patients receiving aprocitentan 25 mg (twice the recommended dose) and 2% on placebo in the clinical trial, requiring additional diuretic use in some patients. Older age and chronic kidney disease are risk factors for edema/fluid retention with TRYVIO. TRYVIO has not been studied in patients with heart failure New York Heart Association stage III–IV, unstable cardiac function, or with NTproBNP ≥500 pg/mL. TRYVIO is not recommended in these patients.
Monitor for signs and symptoms of fluid retention, weight gain, and worsening heart failure. If clinically significant fluid retention develops, treat appropriately, and consider discontinuation of TRYVIO.
Decreases in hemoglobin concentration and hematocrit have occurred following administration of other ERAs and were observed in the clinical trial with TRYVIO. Hemoglobin decreases usually presented early, stabilized thereafter, and were reversible after discontinuation. A decrease in hemoglobin of >2 g/dL from baseline was observed in 7% of patients compared to 1% of placebo patients. A decrease to below 10.0 g/dL was observed in 3% of TRYVIO-treated patients compared to 0 patients taking placebo. Initiation of TRYVIO is not recommended in patients with severe anemia. Measure hemoglobin prior to initiation of treatment and periodically during treatment as clinically indicated.
TRYVIO, like other ERAs, may have an adverse effect on spermatogenesis. Counsel men about potential effects on fertility.
The most common adverse reactions (more frequent than placebo and ≥2% in TRYVIO-treated patients) are edema/fluid retention and anemia.
There are no data on the presence of aprocitentan in human milk, the effects on the breastfed infant, or the effect on milk production. Because of the potential for serious adverse reactions in breastfed infants, advise women not to breastfeed during treatment with TRYVIO.
TRYVIO is not recommended in patients with kidney failure (eGFR <15 mL/min) or on dialysis. Patients with renal impairment are at increased risk of edema/fluid retention.
TRYVIO is not recommended in patients with moderate and severe hepatic impairment (Child-Pugh class B and C) because these patients may be at increased risk for poor outcomes from hepatotoxicity.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088. Please see full Prescribing Information including BOXED Warning and Medication Guide.
TRYVIO is an endothelin receptor antagonist indicated for the treatment of hypertension in combination with other antihypertensive drugs, to lower blood pressure in adult patients who are not adequately controlled on other drugs. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions.